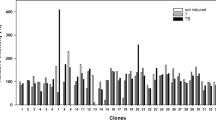
Summary
Recently, a sweet taste receptor family, the T1R family, that recognizes some carbohydrates including sucrose was identified. Although the T1R3 molecule is known to participate in heterodimers that are used as sweet- and umamitasting receptors, there is no evidence that T1R3 alone recognizes similar ligands. We demonstrate for the first time that the candidate sweet taste receptor T1R3 is essential for the recognition and response to the disaccharide trehalose. Our system is a valuable tool not only for understanding the relationship between sweeteners and their receptors but also for exploring the diversities of their receptors, resulting in the design of new high-potency sweeteners.
Similar content being viewed by others
References
Arai, C.; Kohguchi, M.; Akamatsu, S., et al. Trehalose suppresses lipopolysaccharide-induced osteoclastogenesis in mouse bone marrows. Nutr. Res. 21:993–999; 2001.
Ariyasu, T.; Arai, C.; Yoshizane, C., et al. Trehalose augments osteoprotegerin production in the FHs74Int human intestinal epithelial cell line. In Vitro Cell. Dev. Biol. 38A:30–34; 2002.
Baker, E. K.; Colley, N. J.; Zuker, C. S.. The cyclophilin homolog NinaA functions as a chaperone, forming a stable complex in vivo with its protein target rhodopsin. EMBO J. 13:4886–4895; 1994.
Capeless, C. G.; Whitney, G.. The genetic basis of preference for sweet substances among inbred strains of mice: preference ratio phenotypes and the alleles of the Sac and dpa loci. Chem. Senses 20:291–298; 1995.
Chandrashekar, J.; Mueller, K. L.; Hoon, M. A., et al. T2Rs function as bitter taste receptors. Cell 100:703–711; 2000.
Clapham, D. E.. Calcium signaling. Cell 80:259–268; 1995.
Colaco, C.; Sen, S.; Thangavelu, M., et al. Extraordinary stability of enzymes dried in trehalose: simplified molecular biology. Bio/Technology 10:1007–1011; 1992.
Draber, P.; Fraberova, E.; Novakova, M. Stability of monoclonal IgM antibodies freeze-dried in the presence of trehalose. J. Immunol. Methods 181:37–43; 1995.
Dwyer, N. D.; Troemel, E. R.; Sengupta, P., et al. Odorant receptor localization to olfactory cilia is mediated by ODR-4, a novel membrane-associated protein. Cell 93:455–466; 1998.
Elbein, A. D.. The metabolism of α,α-trehalose. In: Tipson, R. S.; Horton, D., ed. Advances in carbohydrate chemistry and biochemistry. Vol. 30. New York: Academic Press; 1974:227–256.
Eroglu, A.; Russo, M. J.; Bieganski, R., et al. Intracellular trehalose improves the survival of cryopreserved mammalian cells. Nat. Biotechnol. 18:163–167; 2000.
Firestein, S. How the olfactory system makes sense of scents. Nature 413:211–218; 2001.
Fuller, J. L. Single-locus control of saccharin preference in mice. J. Hered. 65:33–36; 1974.
Guo, N.; Puhlev, I.; Brown, D. R., et al. Trehalose expression confers desiccation tolerance on human cells. Nat. Biotechnol. 18:168–171; 2000.
Harkins, A. B.; Kurebayashi, N.; Baylor, S. M.. Resting myoplasmic free calcium in frog skeletal muscle fibers estimated with fluo-3. Biophys. J. 65:865–881; 1993.
Ishimoto, H.; Matsumoto, A.; Tanimura, T. Molecular identification of a taste receptor gene for trehalose in Drosophila. Science 289:116–119; 2000.
Kerper, L. E.; Hinkle, P. M. Cellular uptake of lead is activated by depletion of intracellular calcium stores. J. Biol. Chem. 272:8346–8352; 1997.
Kitagawa, M.; Kusakabe, Y.; Miura, H., et al.. Molecular genetic identification of a candidate receptor gene for sweet taste. Biochem. Biophys. Res. Commun. 283:236–242; 2001.
Krautwurst, D.; Yau, K.-W.; Reed, R. R.. Identification of ligands for olfactory receptors by functional expression of a receptor library. Cell 95:917–926; 1998.
Li, X.; Staszewski, L.; Xu, H., et al. Human receptors for sweet and umami taste. Proc. Natl. Acad. Sci. USA 99:4692–4696; 2002.
Lindemann, B. Receptors and transduction in taste. Nature 413:219–225; 2001.
Lush, I. E.. The genetics of tasting in mice VI. Saccharin, acesulfame, dulcin and sucrose. Genet. Res. 53:95–99; 1989.
Max, M.; Shanker, Y. G.; Huang, L., et al. Tas1r3, encoding a new candidate receptor, is allelic to the sweet responsiveness locus Sac. Nat. Genet. 28:58–63; 2001.
Minta, A.; Kao, J. P. Y.; Tsien, R. Y. Fluorescent indicators for cytosolic calcium based on rhodamine and fluorescein chromophores. J. Biol. Chem. 264:8171–8178; 1989.
Mody, S. M.; Ho, M. K.; Joshi, S. A., et al. Incorporation of GαZ-specific sequence at the carboxyl terminus increases the promiscuity of Gα16 toward Gi-coupled receptors. Mol. Pharmacol. 57:13–23; 2000.
Montmayeur, J.-P.; Liberles, S. D.; Matsunami, H., et al. A candidate taste receptor gene near a sweet taste locus. Nat. Neurosci. 4:492–498; 2000.
Nelson, G.; Chandrashekar, J.; Hoon, M. A., et al. An amino-acid taste receptor. Nature 416:199–202; 2002.
Nelson, G.; Hoon, M. A.; Chandrashekar, J., et al. Mammalian sweet taste receptors. Cell 106:381–390; 2001.
Nishizaki, Y.; Yoshizane, C.; Toshimori, Y., et al. Disaccharide-trehalose inhibits bone resorption in ovariectomized mice. Nutr. Res. 20:653–664; 2000.
Offermanns, S.; Simon, M. I. Gα15 and Gα16 couple a wide variety of receptors to phospholipase C. J. Biol. Chem. 270:15175–15180; 1995.
Portmann, M.-O.; Birch, G.. Sweet taste and solution properties of α,α-trehalose. J. Sci. Food Agric. 69:275–281; 1995.
Sainz, E.; Korley, J. N.; Battey, J. F., et al. Identification of a novel member of the T1R family of putative taste receptors. J. Neurochem. 77:896–903; 2001.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ariyasu, T., Matsumoto, S., Kyono, F. et al. Taste receptor T1R3 is an essential molecule for the cellular recognition of the disaccharide trehalose. In Vitro Cell.Dev.Biol.-Animal 39, 80–88 (2003). https://doi.org/10.1290/1543-706X(2003)039<0080:TRTIAE>2.0.CO;2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/1543-706X(2003)039<0080:TRTIAE>2.0.CO;2